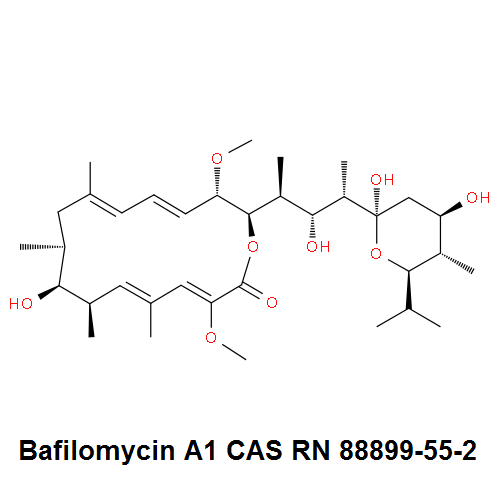
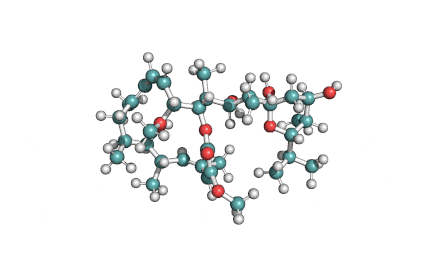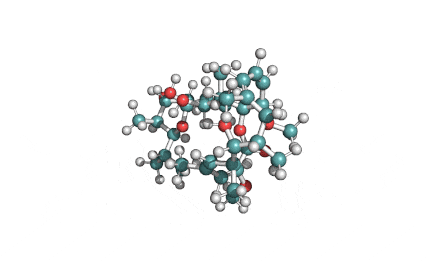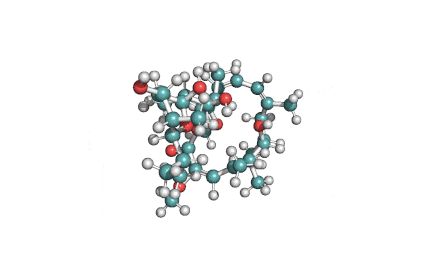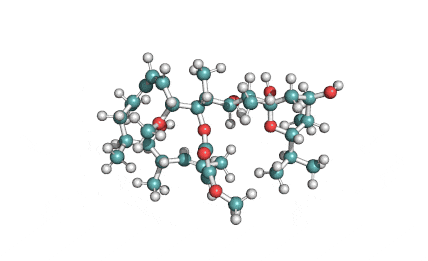
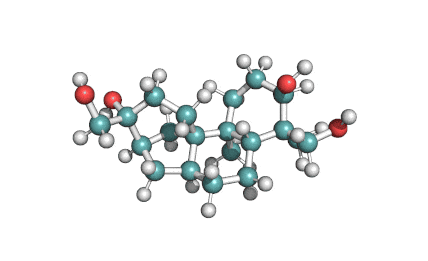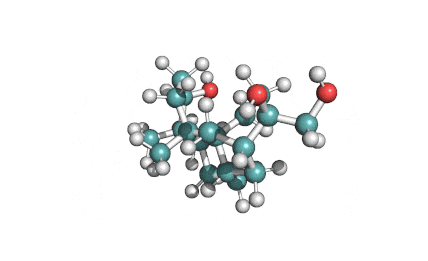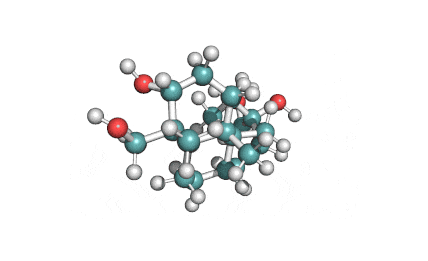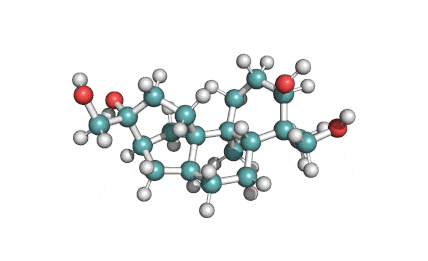
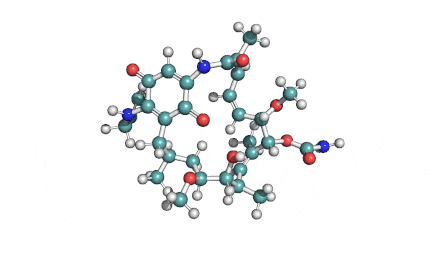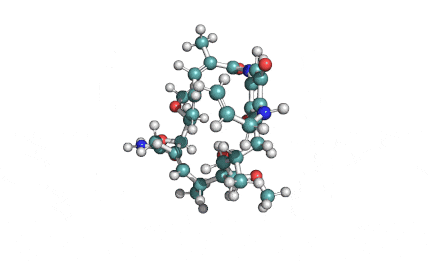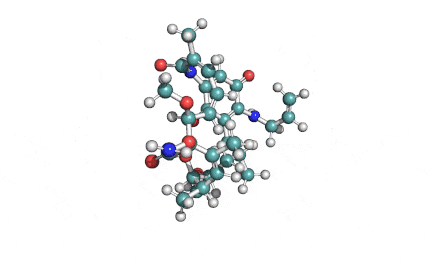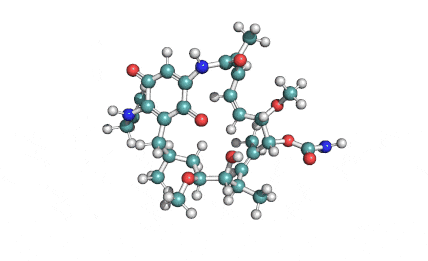
Borrelidin
Details
Specifications
Chemical identification
- Treponemycin
- Cyclopentanecarboxylic acid
- 2-[(4E,6E)-7-Cyano-8,16-dihydroxy-9,11,13,15-tetramethyl-18-oxooxacyclooctadeca-4,6-dien-2-yl]cyclopentanecarboxylic acid
- RTECS ED8750000
Borrelidin is a macrolide-polyketide antibiotic displaying anti-viral, anti-bacterial, anti-malarial, and anti-angiogenic properties.
Further Information
Borrelidin is soluble in DMF, DMSO, Ethanol, Ethyl acetate, Methanol
- Cyclin-dependent kinase (CDK) inhibitor ;
- Antiviral agent
- Anti- Angiogenetic agent
- Apoptosis inducer
Borrelidin, a potent antifungal agent
Composition
Special Info
Other Fields