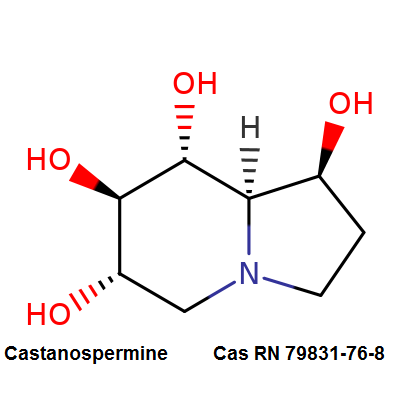
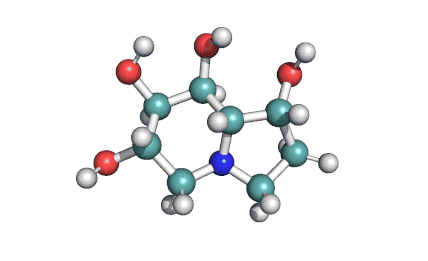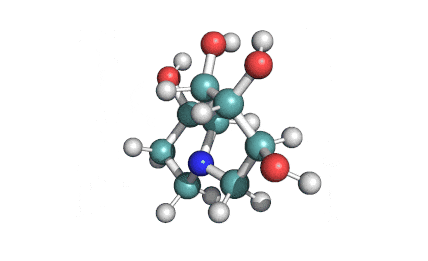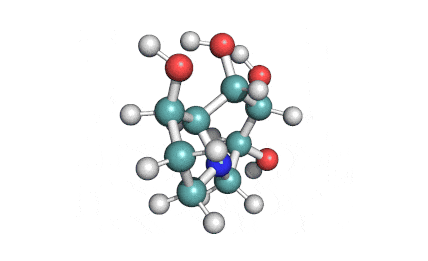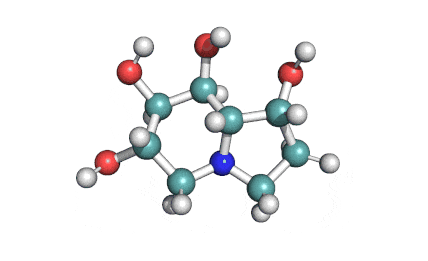
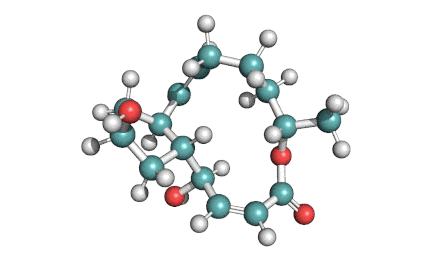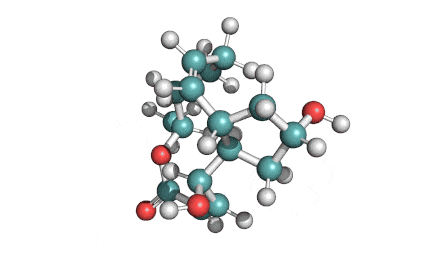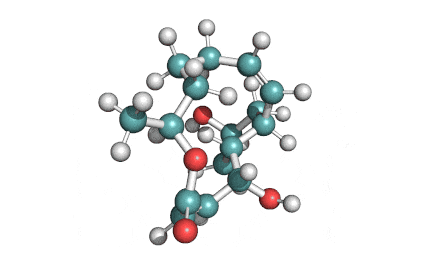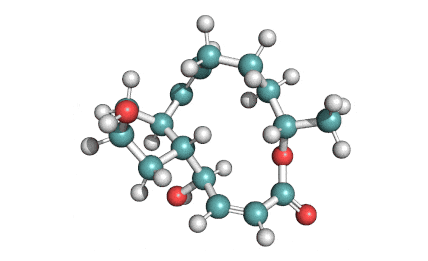
Castanospermine
Details
Specifications
Chemical identification
Synonyms:
- 1,6,7,8-Tetrahydroxyoctahydroindolizine
Alkaloid extracted from an australian legume. Potent inhibitor of α- and β-glucosidase activity in fibroblast extracts; inhibits the glycoprotein processing cycle. Exhibits antiviral properties. Inhibits angiogenesis.
Further Information
Soluble in water.
Indolizidine alkaloid
Glycosidase Inhibitor
Angiogenesis inhibitor
Potent inhibitor of α- and β-glucosidases.
Inhibits HIV infectivity.
Inhibits angiogenesis.
Inhibits thyroglobulin secretion.
Antitumor activty.
Inhibits protein glycosylation.
Composition
Special Info
Other Fields