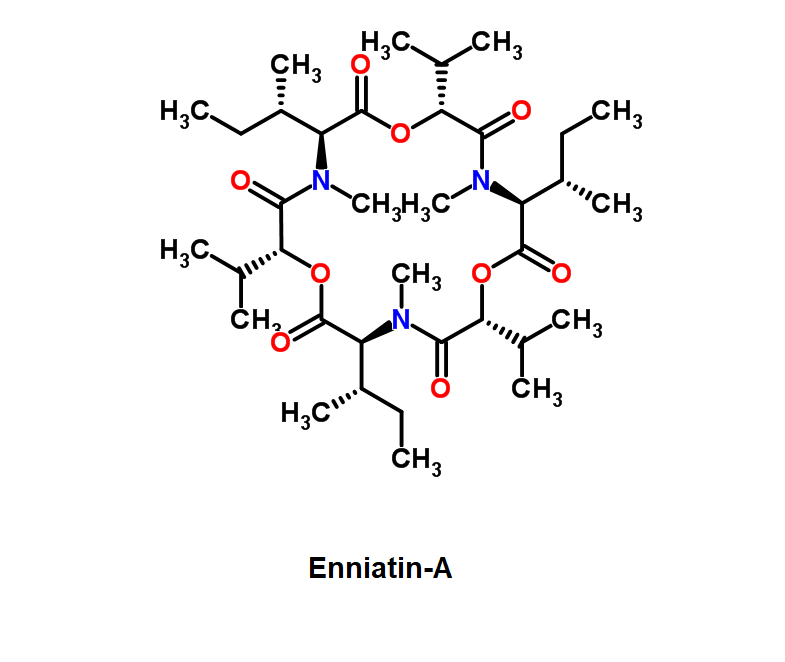
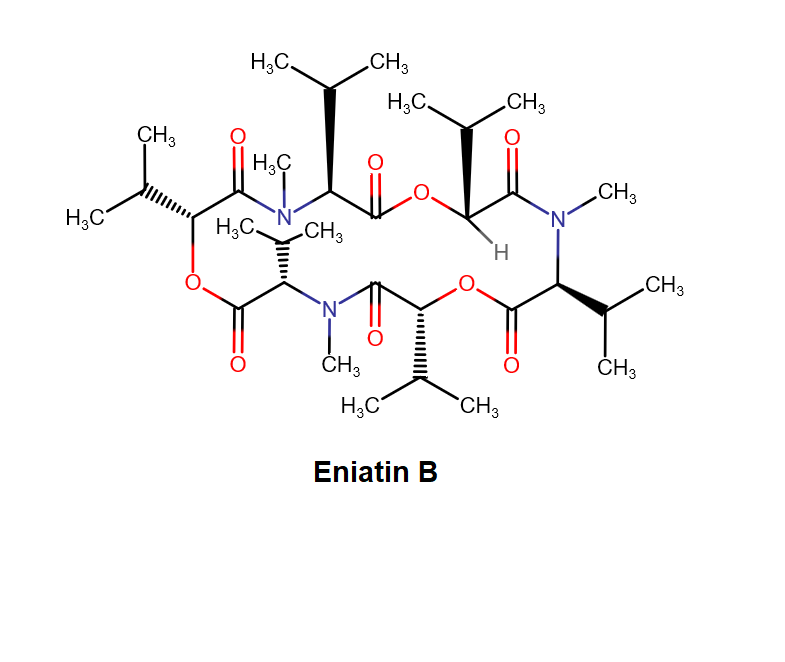
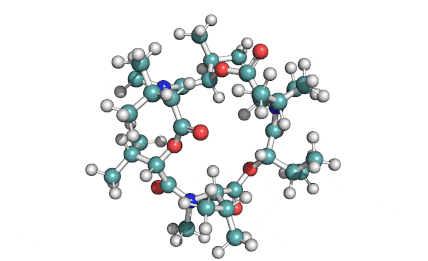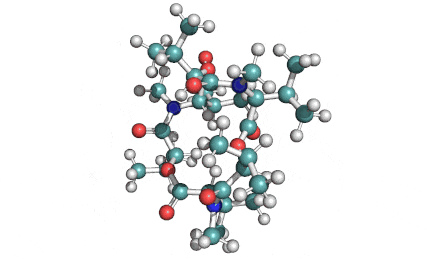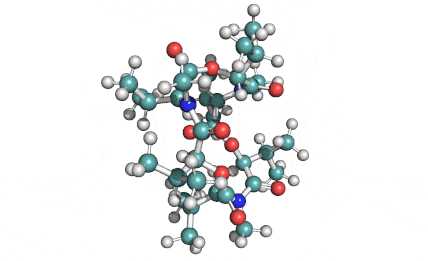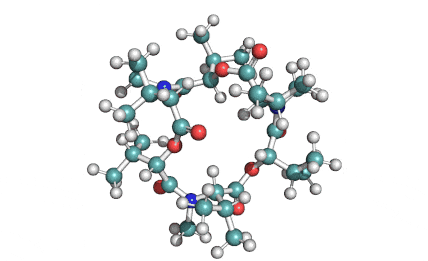
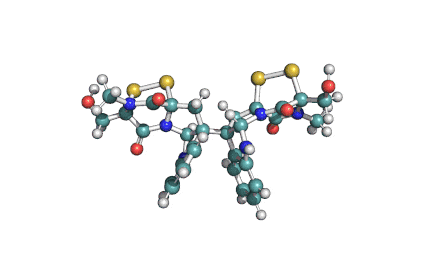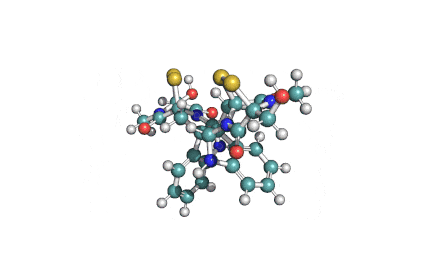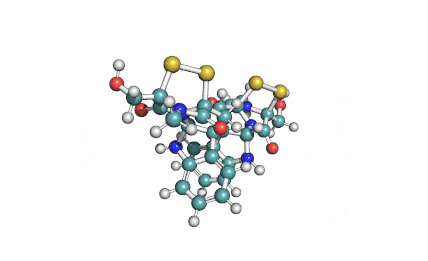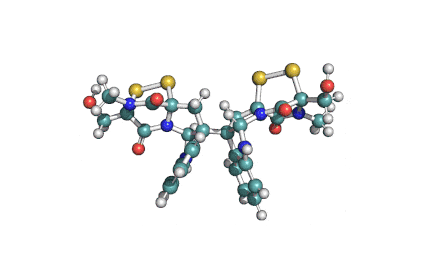
Enniatin A
Details
Specifications
Chemical identification
Synonyms:
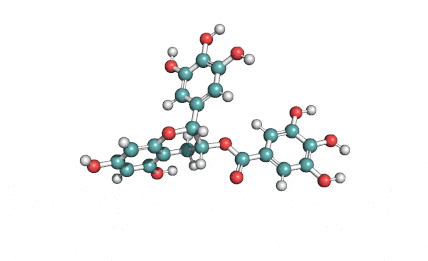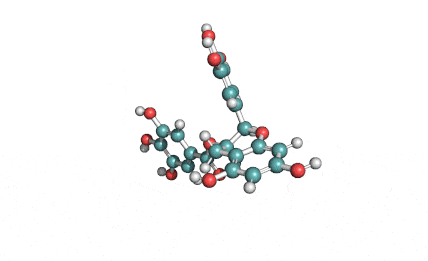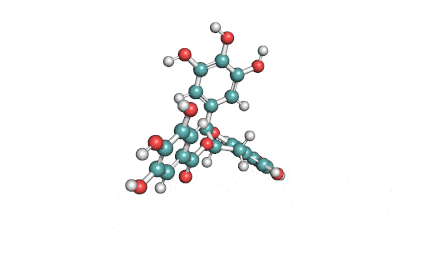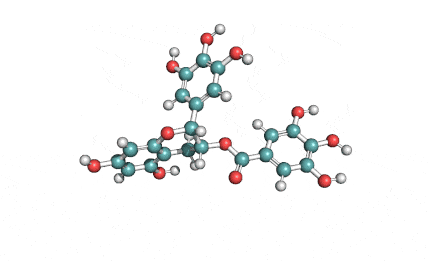
Chemical names: Fusafungine ; Enniatin-A
IUPAC: (3S,9S,12R,15S,18R)-9,15-bis[(2S)-butan-2-yl]-3-[(2R)-butan-2-yl]-4,10,16-trimethyl-6,12,18-tri(propan-2-yl)-1,7,13-trioxa-4,10,16-triazacyclooctadecane-2,5,8,11,14,17-hexone
A Fusarium produced depsipeptide ionophore
RTECS#
Condensed IUPAC: cyclo[N(Me)Ile-D-OVal-N(Me)Ile-DL-OVal-N(Me)aIle-D-OVal]
Enniatin A is a least abundant commponent of the Enniatin-complex. It has been recently made available as a pure compound, thanks to the most advanced preparative HPLC methods.
Further Information
Soluble in ethanol, methanol, DMSO, DMF.
Chemical classification: Cyclic depsipeptide.
Biological classification:
Natural enniatins occure as a mixture of several related analogs, known as "Enniatins", or "Enniatin complex".
Enniatins occure in nature in the mycelia of some Fusarium species. Being cyclic and hydrophopic molecules, enniatins are able to act as ionophores. More recently their effects on acyl-CoA cholesterol transferase, transporters and the selectivity of their antitumor action have received more focus. Enniatin has not previously been available for investigation.
Composition
Special Info
Other Fields