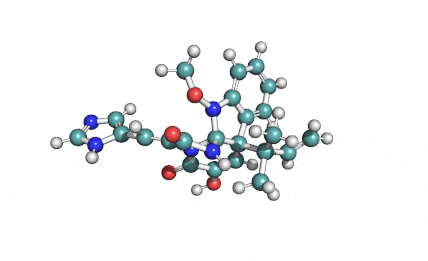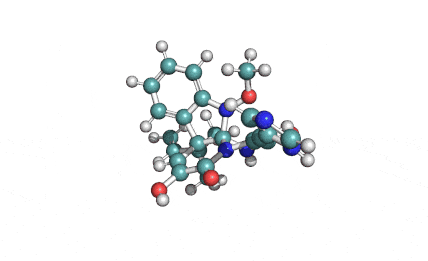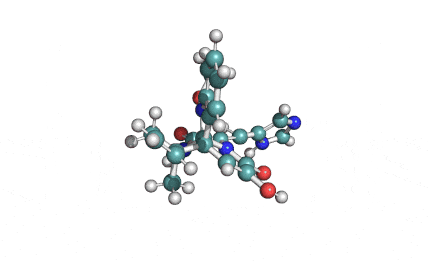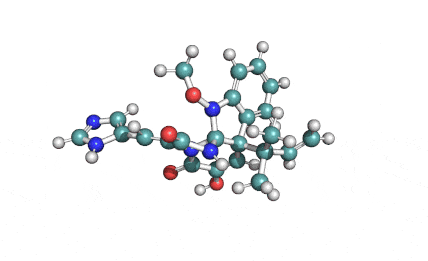
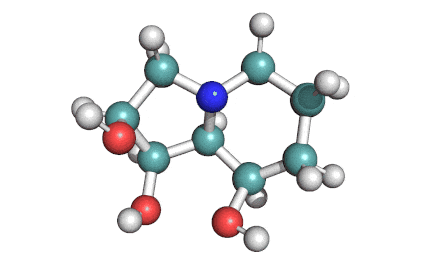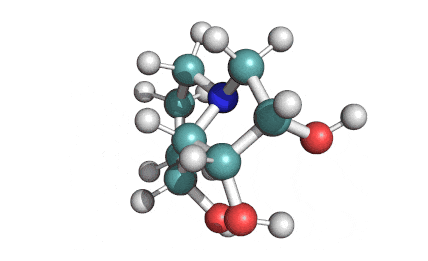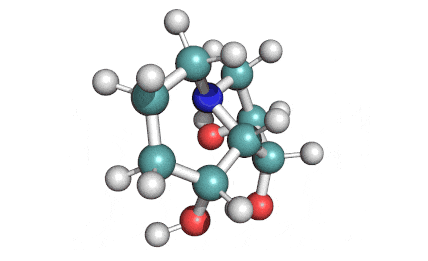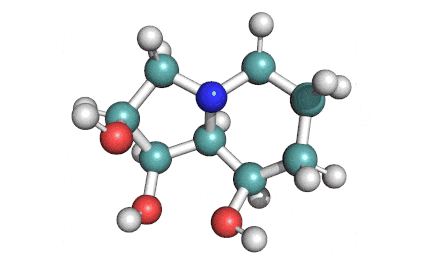
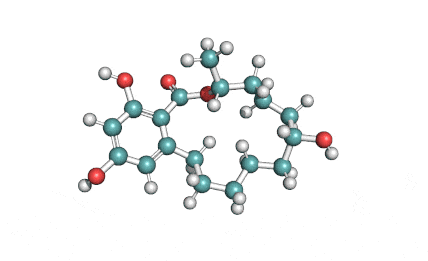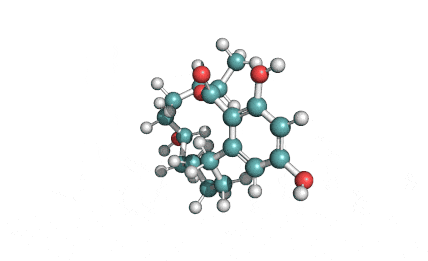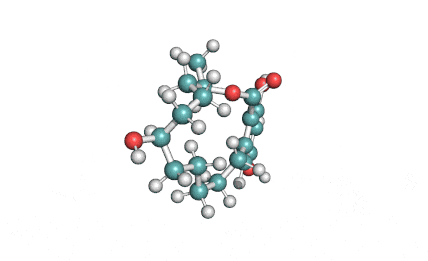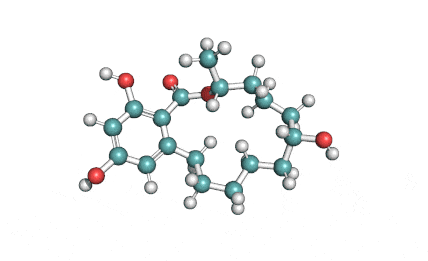
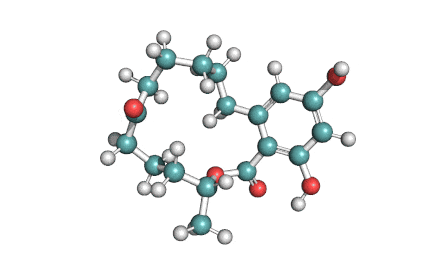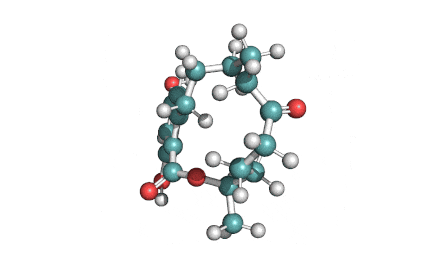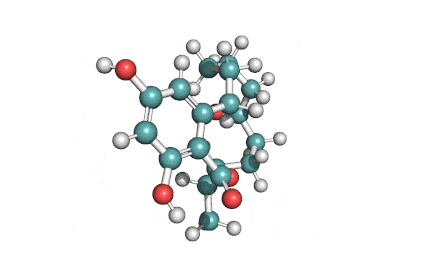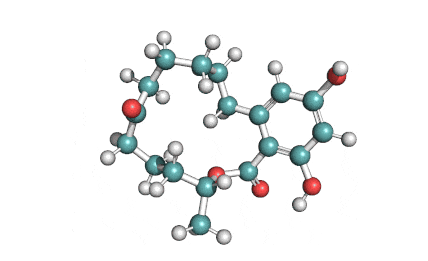
Meleagrin
Details
Specifications
Chemical identification
Synonyms: 6-O-Methyloxaline
Chemical names:
IUPAC: (3E,7aS,12aR)-![]() 6-
6-![]() hydroxy-
hydroxy-![]() 3-
3-![]() (1H-
(1H-![]() imidazol-
imidazol-![]() 4-
4-![]() ylmethylidene)-
ylmethylidene)-![]() 12-
12-![]() methoxy-
methoxy-![]() 7a-
7a-![]() (2-
(2-![]() methylbut-
methylbut-![]() 3-
3-![]() en-
en-![]() 2-
2-![]() yl)-
yl)-![]() 7a,12-
7a,12-![]() dihydro-
dihydro-![]() 1H,5H-
1H,5H-![]() imidazo[1',2':1,2]pyrido[2,3-
imidazo[1',2':1,2]pyrido[2,3-![]() b]indole-
b]indole-![]() 2,5(3H)-
2,5(3H)-![]() dione
dione
RTECS#
Meleagrin, a fungal alcaloide antibiotic, posesses antitumor and antibiotic activity. Meleagrin has been identified as an inhibitor of the bacterial enoyl-acyl carrier protein reductase.
Meleagrin, CAS 71751-77-4, the fungal alcaloide, should not be confused with the low molecular weight peptide cas 121272-63-7, which is isolated from turkey also called meleagrin.
Further Information
Ethanol, methanol, DMF or DMSO. Poor water solubility
Alcaloide antibiotic
Inhibitor of: Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
Composition
Special Info
Other Fields