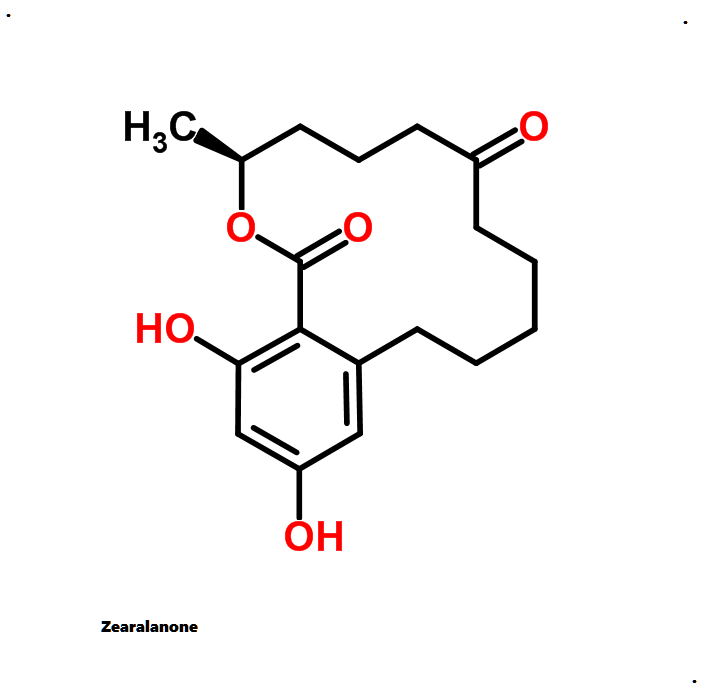
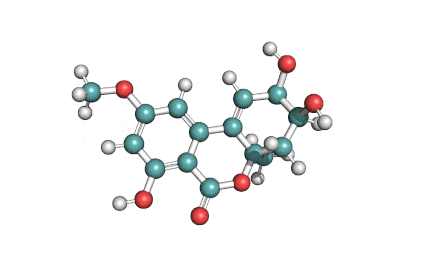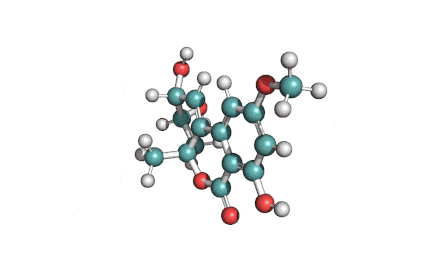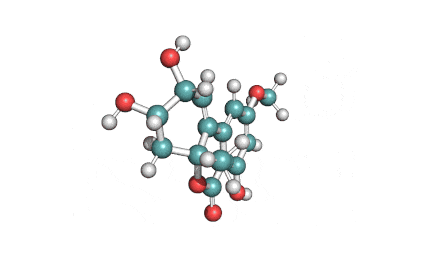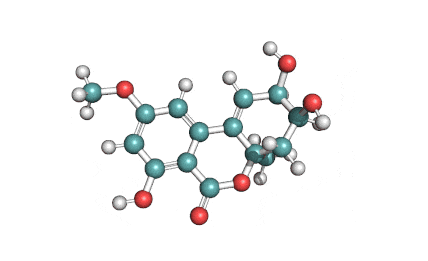
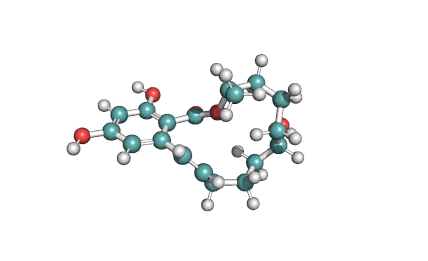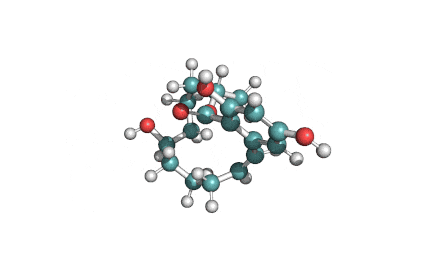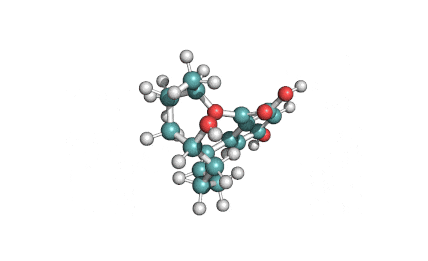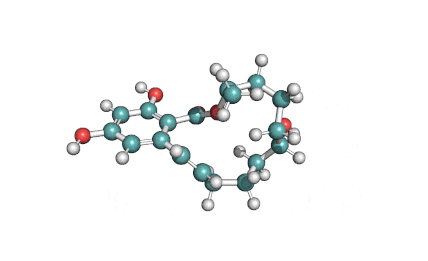
Zearalanone
Details
Specifications
Chemical identification
Synonyms: Zearalanone; (S)-Zearalanone; Zanone
Chemical names:
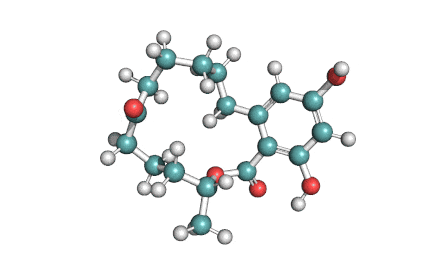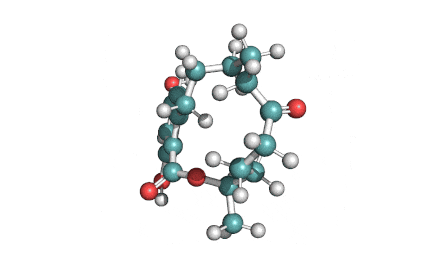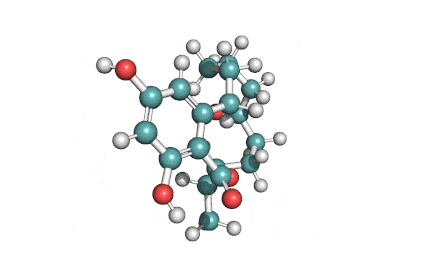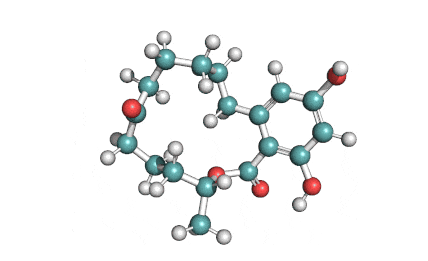
IUPAC: (11S)-15,17-dihydroxy-11-methyl-12-oxabicyclo[12.4.0]octadeca-1(18),14,16-triene-7,13-dione
RTECS# DM2530000
Zearalanone is a minor component of the zearalenone complex produced by several species of Fusarium. Like the more abundant analogues, zearalanone causes estrogenic effects in domestic livestock. Zearalanone is a metabolite of α-zearalanol, a commercially available growth promotant in animals, and is a useful standard for detection of zearanol-contaminated products and Fusarium-contaminated grains.
Further Information
Soluble in ethanol, methanol, DMF or DMSO.
Soluble scarcely in water.
- Potential anti-tumor agent.
- Estrogen receptor agonist
- Androgen receptor antagonist
Composition
Other Fields