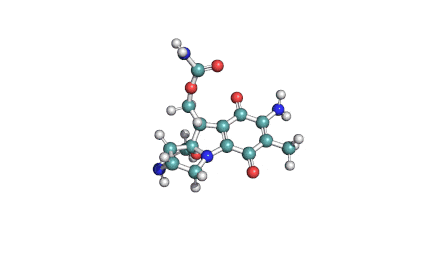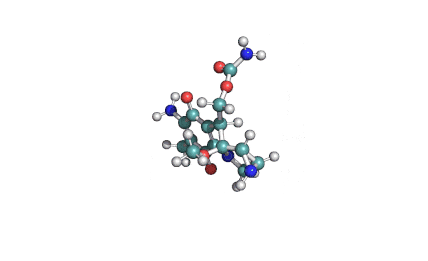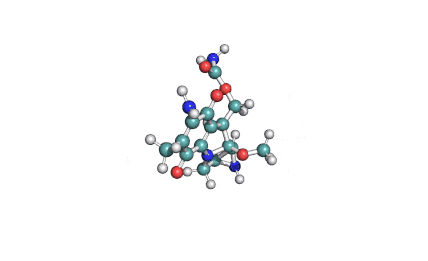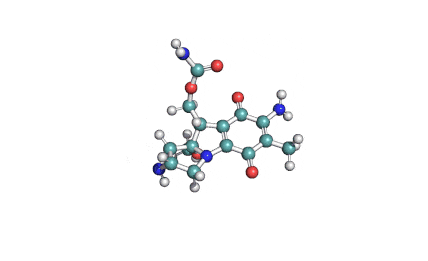
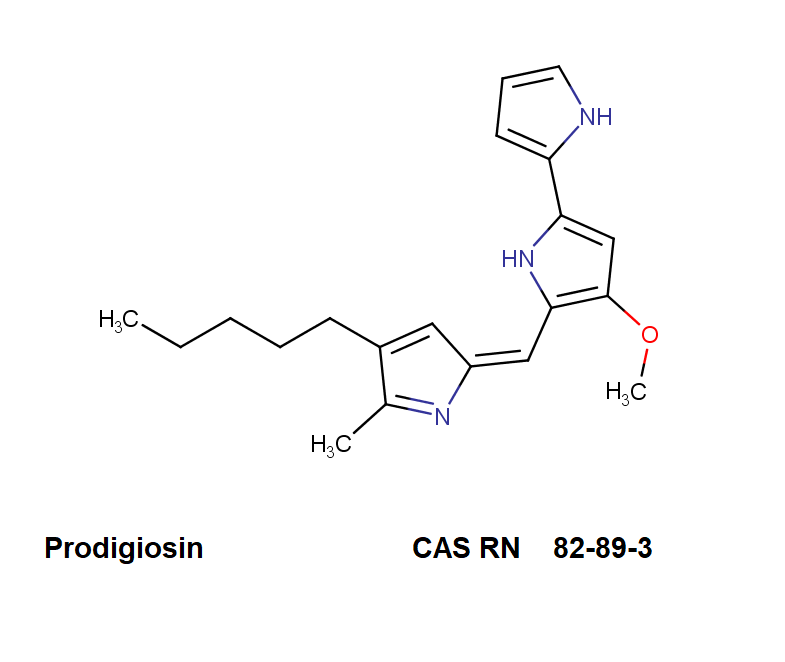
Ibotenic acid
Details
Specifications
Chemical identification
Synonyms:
▪ Ibotenic acid
▪ Pramuscimol
▪ 5-Isoxazoleacetic acid, alpha-amino-2,3-dihydro-3-oxo-
▪ α-Amino-3-hydroxy-5-isoxazoleacetic acid
▪ (S)-2-amino-2-(3-oxo-2,3-dihydroisoxazol-5-yl)acetic acid
Chemical names:
IUPAC:
Amino(3-oxo-2,3-dihydro-1,2-oxazol-5-yl)acetic acid
RTECS# NY2100000
Further Information
Composition
Supply related information
Special Info
Other Fields



![Mitomycin C [1mg Mitomycin C + 24mg NaCl] Mitomycin C [1mg Mitomycin C + 24mg NaCl]](/sites/default/files/2024-02/Mitomycin-C%20NaCL%20Mix.png)