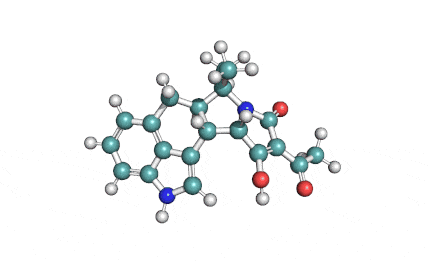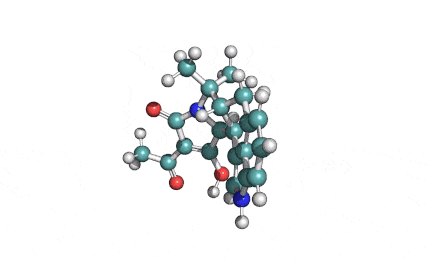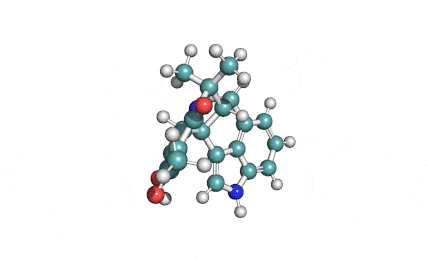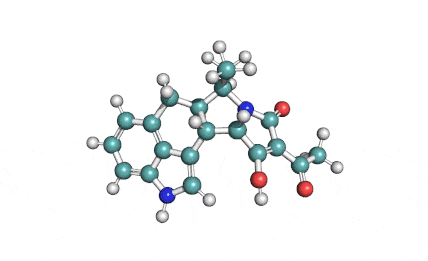
K252a
Details
Specifications
Chemical identification
RTECS NZ0550000
K252a: Cell permeable protein kinase inhibitor
Further Information
Readily soluble in chloroform, acetonitrile, acetone, dioxane, tetrahydrofuran, pyridine;soluble in ethanol, methanol, 1-propanol, ethyl acetate and n-butanol; insoluble in water, 2-propanol
- indolocarbazole alkaloid
- PK inhibitor
K252a is a highly potent cell permeable inhibitor of CaM kinase and phosphorylase kinase . At high concentrations it also inhibitors of serine/threonine protein kinases. K252a inhibits tyrosine phosphorylation of Trk A induced by NGF K252a is reported to promote myogenic differentiation in C2 mouse myoblasts
Composition
Other Fields