Nivalenol
Molecular Formula
C15H20O7
M.W.
312.32
CAS number
23282-20-4
Details
Source
Semisynthetic
Fermentek product Code
NIV-001
Brand/grade
For research
Specifications
Appearance
White to Off-White powder
Purity by HPLC
≥98% ; refer to CoA for more data
Purity By TLC
≥98% ; refer to CoA for more data
Solubility test
Clear colorless solution at 10 mg/ml Methanol or DMSO
Chemical identification
Names and identifiers
IUPAC Name
(3β,4α,7α,12ξ)-3,4,7,15-Tetrahydroxy-12,13-epoxytrichothec-9-en-8-one
RTECS#
YD0167000
EU number
621-749-5
Description
Nivalenol is a type - B trichothecene mycotoxin produced by various Fusarium species, such as Fusarium cerealis, Fusarium graminearum, Fusarium nivale. Besides the natural toxin produced directly by molds, Nivalenol may be also formed by higher plants and by mammals, as a product of biodegradation of other, related thrichothecenes.
InChl Key
UKOTXHQERFPCBU-XLYGLCRNSA-N
Canonical SMILES
CC1=CC2C(C(C1=O)O)(C3(C(C(C(C34CO4)O2)O)O)C)CO
Isomeric SMILES
CC1=C[C@@H]2[C@]([C@@H](C1=O)O)([C@]3([C@@H]([C@H]([C@H](C34CO4)O2)O)O)C)CO
Further Information
Solubility ( literature )
Dichloromethane, DMSO, Methanol. Slightly soluble in water
Compound Classification
Chemical class:
Trichothecene mycotoxin
Storage, handling
Store in a freezer upon arrival, at -10°C to -25°C
Use the original container to store the product.
Keep the lid tightly closed.
trichocethenes are thermostable compounds and are not destroyed in boiling.
Retest time
3 Years
Applications
Nivalenol (0.01 mg/ml) induces apoptosis in HL60 cells (Ueno et al., 1995). Nivalenol was shown to inhibit protein synthesis in rabbit reticulocytes in vitro. Nivalenol inhibited the synthesis of nucleic acids in vitro.
Disclaimer
For Research use only
Not for Human or Drug use
Not extracted from humans or animals
Refer to MSDS for further safety and handling instructions
Semi-Synthetic
Composition
Ingredient type
Fermentek product
Special Info
Available since
Other Fields
Fermentek Product Category
Signal to sort
N
Image
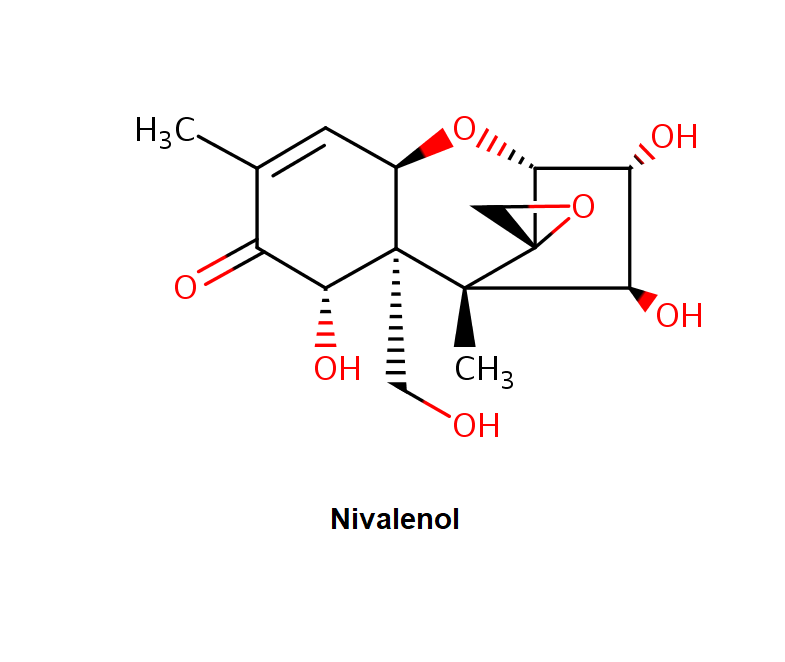
Image
Image

Image